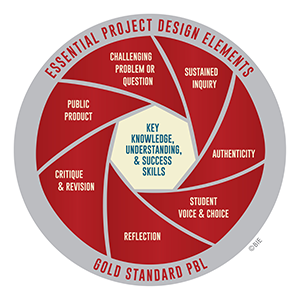
 My formal training in PBL is was through Buck Institute's Gold Standard PBL 101 workshop. My plan is to use the next series of posts to take you through the essential project design elements for my first PBL project in my general chemistry classes. Feel free to comment with suggestions.
The first step of this PBL project was the challenging problem or question, for my students the guiding question was: How can we sold the problem of plastics in the ocean/freshwater. Typically for Gold Standard PBL, the question needs to be a problem to solve or a question to answer. Our entry event into this project was having my students watch two videos: 1. Midway, a Plastic island http://www.cnn.com/videos/us/2016/11/30/midway-plastic-island-nick-paton-walsh-orig-jql.cnn and 2. Beads of destruction https://youtu.be/o0NikCMZCFE?t=4s Both of these videos are very powerful and created a great hook for kids that led them into the driving question. From the driving question, in groups students brainstormed need to know questions about plastics. Underlying all of this was some of the basic chemistry of plastics including arrangement of elements on the periodic table and how that arrangement leads to different types of bonds and bonding which can be used in material science (NGSS, HS-PS1-2). For authenticity, even though Atwater, Ca is roughly 100 miles from the ocean, within 5 miles of our school is the Merced River. The Merced is a river that tumbles out of the high Sierras, runs through Yosemite national park, and drops into the central valley where it ultimately joins the San Joaquin river. The San Joaquin flows into San Francisco bay and then the Pacific Ocean. Many of my students have been to the river, either fishing or just floating down the river on innertubes. The river feeds Lake Yosemite in nearby Merced, which again, many of my students have spent afternoons swimming or fishing or just having a family barbeque on the grassy banks. With this, students can identify with the the effects of plastic pollution on wildlife and began to see how microplastics could possibly harm them.
2 Comments
Isabella Bruyere is 10th grader, this is an important read, I feel one of the most important thoughts is: So how did my love for school change? Simple: school stopped being about learning. As I entered high school, and even middle school, everyone around me, teachers and students alike, had the mindset of “cram cram cram, A’s, A’s A’s”. They’ll shove useless information into your head as fast as possible, “it’s okay if you don’t understand it, just memorize it and get an A on your exam!” The exam? An hour in a room of no talking, just bubbling in multiple choice answers while bubbles of anxiety grew in your stomach. School slowly became a place of memorizing facts just long enough to get the A, doing the bare minimum to get into the best college. Everything was just to get into college, to be better than your peers. Why help your classmate? Why not sabotage them so you have less people to compete with when it comes to applying to Harvard, Stanford, Yale. That is the mentality that I hate, yet it is the mentality of everyone around me, and maybe even myself. Read the rest of this here. What do you think?
Not long after I became interested in project based learning, I read Tony Wagner and Ted Dintersmith's book, Most Likey to Succeed. The book was also turned in to a fantastic documentary. I highly recommend both (as always, read the book first, just saying). After reading about this study, I began questioning the need to keep doing things as we have always done in education:
The Lawrenceville School is consistently rated as one of the very best U.S. elite private high schools (according to Forbes). A decade ago, it ran fascinating experiment with students taking core science courses. When students returned after summer vacation, they were asked to retake the final exam they had completed three months earlier. Actually, it was a simplified version of the final, as the faculty eliminated any detailed questions that students should't be expected to remember a few months later. The results were stunning. When students took the final in June, the average grade was a B+ (87%); when the simplified test was taken in September, the average grade was an F (58%). Not one student retained mastery of all important concepts covered by the course. Following this experiment, Lawrenceville completely rethought the way courses were taught, eliminating almost half of the content to emphasize deeper learning. When repeating the experiment in subsequent years, the results were far more satisfactory.
As I began to think about project based learning, I became worried that I would never get through all of the curriculum needed. How could I do long term projects that required kids to research topics and be exposed to inquiry methods of teaching. These both would take time and I only have approximately 184 days with students. After reading the study done at Lawrenceville, I can better about doing less chemistry, but diving deeper into chemistry. Having students just memorize and regurgitate information for an exam, only to immediately forget this information, does not bode well for education in the U.S. Does this mean I no longer teach chemistry, not at all. My students still do labs, they still listen to lectures and take notes, they still work problems, but I am trying to incorporate more authentic experiences and less multiple choice tests.
The traditional teaching and learning of chemistry has been unchanged for over 100 years. Students sitting in a row, periodic table in hand, listening to a teacher lecture about how to calculate molar mass or work a stoichiometry problem or balance an equation. I've been doing this for 20 years. A few years ago my district went 1:1, now every student in our district now has a Chromebook checked out to them. Pew Research Center has found as of 2013, 78% of teens 12-17 have a cell phone, I would guess that number is higher now. Students now have immediate access to anything that Google can provide them.
On a recent lesson, I had students draw Lewis structures of molecules. As I bounced from student to student checking their structures I notice that quite a few of my 1st period students we drawing Lewis structures different from how I had showed them, the structures were technically correct but kids would not have known this unless taught by another teacher or if they looked them up. What a percentage of my students were doing was just to simply Google: Lewis structure CO2. Kids can now do this with a large % of basic chemistry concepts. I have found online balanced equation tools as well as online stoichiometry tools. Personally I have used Siri to quickly give me the molar mass of a compound when preparing reagents for a lab. I am not planning on abandoning basic chemistry concepts, I just do not plan on spending as much time on concepts that can easily be looked up. I often hear fellow teachers argue that we should not allow kids to use their Chromebook or phone in class, but I don't feel this is a reflection of the real world. A person working in industry will use any or all of the available tools including the internet, and of course, we should be getting kids ready for the real world of work. So what is answer? For me, it is to start asking questions that can not be simply Googled. Three years ago, I stared at a CUE national conference schedule trying to decide what sessions to attend for the day. Project Based Learning in the math class looked interesting. Picking this session to attend would start to change the fundamental way I looked at education, teaching, and learning. At this same conference I discovered the power of Twitter in creating a professional learning network (PLN). I started searching for PBL hashtags (# pbl, #pblchat) which eventually led me to Buck Institute and last summer I had the opportunity to attend Buck's PBL world in Napa, Ca. In the 2017-18 school year, I launched my first true PBL project, more on this project later. My students just finished their first true PBL project, more on this project soon.
|
AuthorI am Jamie Camp, a chemistry teacher from Atwater, Ca. I teach chemistry and A.P chemistry in Atwater, Ca. I blog about the experiences of a PBL chemistry classroom. Archives
December 2017
CategoriesLinks |
||||||
 RSS Feed
RSS Feed
